To receive ongoing notifications about clinical drug trials and patient enrollment, please register in our Cavernous Malformation Registry.
Treatments in Development
Medicines for CCM may be developed with a variety of purposes in mind. For CCM, we need treatments that will:
- Stabilize blood vessels to reduce oozing and the risk of hemorrhage
- Prevent re-hemorrhage after an acute hemorrhage
- Shrink lesions or remove them without surgery
- Prevent lesions from coming back after surgery
- Remove iron deposits in the brain and spinal cord left behind by hemorrhage
- Prevent systemic effects of familial CCM
- Stop familial CCM from being passed to the next generation
Each potential treatment in the pipeline may address one or more of these treatment needs.
At least initially, until there is a cure, some medicines may be more appropriate for some people with the illness than others. For example, a specific medicine might be best suited for those with familial CCM, or, conversely, for those with sporadic CCM. Preferences and tolerances may also play a role. Some people may be more sensitive to side effects. Or, some may want or need a medicine that is used for just a short time while others may want ongoing protection that requires staying on a medicine for a longer time. We will need many choices.
CCM Treatment pipeline
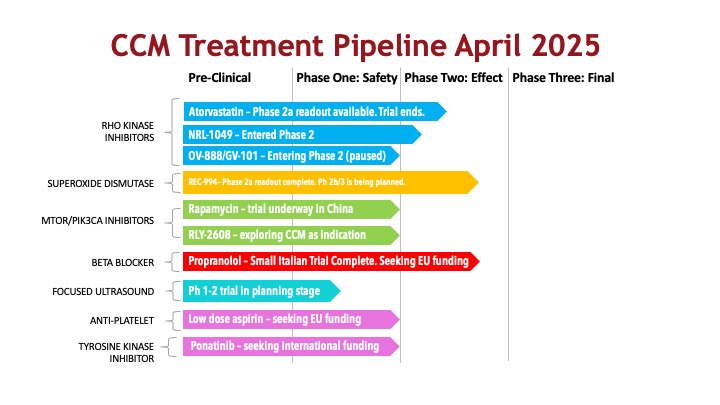
Please see Clinical Trials FAQ for an explanation of clinical trial phases.
Detailed information about treatment classes
Rho Kinase Inhibitors
Rho Kinase and its associated signaling molecules regulate important cellular processes including proliferation and maintaining/changing cell shape. Drugs targeting the Rho Kinase protein are of interest for CCM treatment because in CCM gene mutations cause overactivity of Rho Kinase. Studies in mice have shown that inhibiting (blocking) Rho Kinase function can fix leaky blood vessels and limit the formation of new lesions. Several Rho Kinase inhibitors are of interest to CCM researchers. The statin drug atorvastatin has completed a Phase 1/2 experimental proof of concept trial at the University of Chicago. The medicine did not demonstrate a statistically significant effect on preventing hemorrhage in lesions that had prior hemorrhaged. Other more targeted Rho Kinase inhibitors are in development specifically for CCM.
Oxidative Stress
Oxidative stress refers to a state in which the cell has an overabundance of reactive oxygen species (ROS). ROS are naturally occurring molecules that need to be kept at a regulated level to avoid damaging the cell. A variety of molecules, including REC-994, in development by Recursion, can detoxify ROS. Vitamin D’s effect on CCM lesions may also result from its anti-oxidant properties.
mTOR/PIK3CA inhibition
Several studies have indicated that overexpression of a protein called PIK3CA in a cavernous malformation can cause the lesion to grow aggressively. Researchers are exploring whether inhibiting PIK3CA or the mTOR pathway, which impacts PIK3CA can prevent the growth of lesions. A clinical trial of rapamycin, an mTOR inhibitor has begun in China, and another is seeking funding in the US. Additionally, an exploration of new and repurposed PIK3CA inhibitors for CCM is ongoing with small pilot studies.
Beta Blocker
Propranolol falls into a class of cardiac drugs called beta blockers. They are used to lower blood pressure, manage heart attacks, and improve blood flow. The results of an Italian study, Treat_CCM, were published in 2023. The study was small and open-label (no placebo group), but it seemed to indicate an impact on the development of new cavernous malformations. Subsequent research in zebrafish and mice demonstrated the same effect. This program has not moved forward due to a lack of funding, but there is industry interest in finding ways to bring propranolol to CCM patients with regulatory approval.
Focused Ultrasound
Low-intensity focused ultrasound (LIFU) has been studied for the treatment of CCM and has had impressive results with animal models. The Alliance to Cure Cavernous Malformation and the University of Virginia are working together to launch a Phase 1/2 trial with our patients.
Anti-platelet Agents
Based on retrospective studies of CCM patients who have used anti-thrombotics, some researchers now believe that low-dose aspirin may improve outcomes for CCM patients by reducing the risk of hemorrhage. The theory is that low-dose aspirin prevents clotting, which is suspected to be a trigger for hemorrhage, and also reduces inflammation, a known trigger. An effort to fund an aspirin trial is underway in the United Kingdom.
Tyrosine Kinase Inhibitor
Animal model research has indicated that a tyrosine kinase inhibitor called ponatinib appears to prevent the development of new cavernous malformations and the growth of existing lesions. However, this drug is used in cancer and has many side effects. While researchers are seeking funding for a ponatinib trial, the Alliance to Cure Cavernous Malformation is also working with researchers to develop targeted delivery of medicines directly to cavernous malformation lesions.
Updated 3.31.2025


